Cell and gene therapies (CGTs) have heralded a new era of medicine, promising curative treatments for patients with chronic and often fatal diseases. However, access to these therapies has not come without hindrance. At its core, restricted access to CGTs comes down to a discrepancy between the high prices set by pharma and the price that healthcare systems are willing to pay. While some, such as Novartis’ Zolgensma and Kite’s Yescarta, have successfully (if somewhat partially) navigated this by combining market-level pricing flexibility with risk-sharing schemes, evermore CGTs are steaming towards approval with significant challenges for adoption remaining.
In the current healthcare environment, the majority of emerging CGTs will struggle to establish a foothold, as it is simply not designed to accommodate this step-change in innovation. To stimulate access, pharma and healthcare systems must find common ground across three layers: the evidence required to support CGTs, the value recognition of these innovative therapies, and the pricing models accepted. In this paper, we have explored concerted motions that pharma and healthcare systems can action to unify their thinking across these layers to co-create win-win situations and open access to more patients globally.
The emergence of cell and gene therapies (CGTs) marks a paradigm shift in treatment approaches, where diseases from rare genetic disorders to cancer are all in line for curative potential. The pharmaceutical industry is moving fast to bring these new innovative therapies to market; however, challenges remain around their potential adoption within healthcare systems.
The challenge surrounding reimbursement of CGTs in the current payer landscape is due to three main factors: evidence, risk, and pricing. So far, pharma has been factoring in high manufacturing costs, curative potential, and small patient populations to CGTs prices to support sufficient ROI. The consequence of small patient populations is the reduced scalability of supporting evidence, with pivotal data for CGTs often being generated from small trial cohorts. Further, the novelty of these therapies means that long-term data to substantiate pharma’s curative claims is limited. Through the lens of the payer, this translates to increased risk, where they must pay a high price for a therapy that does not substantiate the curative claims. Additionally, there is a lack of robust real-world evidence (RWE) that can support long term cost-saving claims over a patient’s lifetime.
In the context of pricing and reimbursement (P&R) negotiations, pharma and healthcare systems must attempt to drive towards a middle ground in which the risk/reward is mutually beneficial (Figure 1). Arriving at this middle ground is the sole determinant for patients benefiting from these innovative treatments; ensuring this journey is as short and smooth as possible is of prime importance.
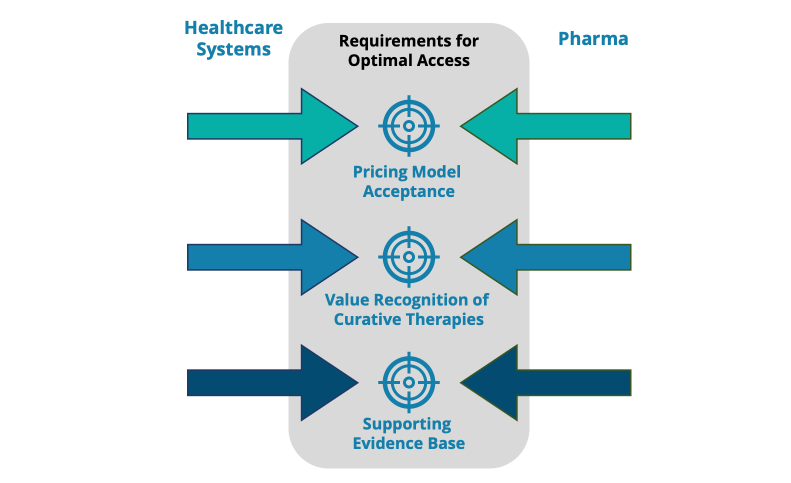
Figure 1: Model for optimal CGT access across the three layers of evidence, value, and price. Pharma & healthcare systems must agree at each level to reach a mutually beneficial middle ground to ensure CGTs access is unlocked for patients to benefit.
These dynamics raise several questions that will define the future of CGT access:
- Is there a ceiling to the price healthcare systems are willing to pay for curative treatments?
- What policy changes are required to support the uptake of CGTs in broader indications?
- What supporting evidence base is needed to support curative claims?
- How can CGT manufacturers lower costs & drive efficiencies in getting GCTs to market at lower prices?
Before looking ahead to the future, it is prudent to examine the current state of CGT access, where the European launch of Bluebird Bio’s β thalassemia treatment, Zynteglo, illustrated an attempt to navigate these issues. BlueBird Bio hoped healthcare systems would value Zynteglo at $1.8M per patient based on lifetime cost-saving claims. Following approval in 2019, Bluebird placed risk-sharing at the forefront of its pricing strategy, with risk-sharing acting as an offset to the high list price. Payers would make an initial payment followed by annual milestones if the treatment continued to provide benefits. In theory, this lowers the up-front cost for healthcare systems while ensuring pharma has skin in the game based on the performance of their product. According to Bluebird’s CEO, Nick Leschly, the biotech was willing to put 80% of its price down to risk-sharing. However, HTA-driven markets across the EU5 rejected this model, finding Zynteglo not cost-effective and demanding steep discounts. The unwillingness of either party to compromise on their demands meant that a favorable middle ground was never reached, leaving patients without access to this new innovative treatment. As a result, Bluebird has since wound down European operations and is proceeding to withdraw marketing authorization for Zynteglo in the region. Europe’s hostility towards Zynteglo has set a restrictive precedent for future innovative curative medicines, with Bluebird’s recently approved cerebral adrenoleukodystrophy treatment Skysona, priced at $3M, planned for launch only in the US. Will this mark the start of a trend not just for Bluebird but for all CGT players aspiring to enter the EU market?
“Finding common ground between pharma and healthcare systems is essential to unlocking access and the true potential of CGTs for patients”
This is just one example of the misalignment between pricing and payer expectations hindering patient access to innovative therapies. If this trend continues, there will be detrimental effects on pharma & healthcare systems across global markets. Within markets where access is restricted, this can lead to limited physician expertise with new CGT treatments and deprioritization for future launches. Globally, pharma will have less incentive to invest in CGTs, leading to a growing disparity in treatment approaches across markets for these new innovative medicines. Healthcare systems worldwide have struggled to cope with the high prices that pharma demand, with Europe’s less centralized reimbursement landscape further compounding this problem. For example, Spain is pushing back against high prices and hindering access to a third of approved CGTs, yet the UK has a greater appetite and stands to reimburse more than 80% of them. We also see differences in pricing policy across major markets. In Germany, there is little appetite for longer-term performance-based contracts, with decision-makers preferring the fiscal simplicity of deep discounts from the outset. Meanwhile, the Inflation Reduction Act has been signed into law in the US, protecting rare disease drugs from price negotiations (1).
Alternative Pricing Models That Could Provide Solutions
The route forward is clear: payers and pharma must find solutions together, and a crucial part of this is co-creating pricing models where both parties win. To be effective, these innovative pricing models must capture the holistic long-term cost savings that CGTs can provide to the healthcare system, which should include; treatment, caregiver, hospitalization, and socio-economic costs associated with the diseases they target. However, as seen in the case of Zynteglo, many payers take a short-sighted view and dismiss these downstream savings.
Consequently, innovation in pricing models require flexibility from both pharma and payers and recognition that there is no one-size fits all strategy. Instead, successful pricing strategies should be tailored to the product, market, and manufacturer to ensure that all stakeholders win. This approach would enable equitable access to novel therapeutics, ultimately benefitting the patient. To date, numerous innovating pricing strategies have been utilized to facilitate access to CGTs (Table 1).
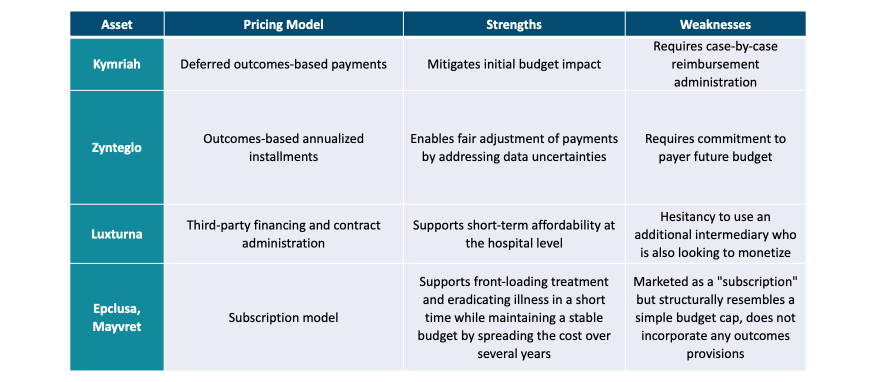
Table 1: Examples of pricing strategies utilized by curative therapies (2)
Novartis demonstrated a keen awareness of these stakeholder interactions across markets in their access strategy for Zolgensma, a curative SMA therapy currently sold in the US market with a list price of $2.12M. The treatment has continued to set new pricing precedents globally, receiving the highest-ever reimbursement price in Japan, successfully negotiating performance-based contracts in Germany, and initiating a multinational HTA assessment with Benelux. The true winners are the patients, but the question remains, how did Novartis succeed where others have failed?
Novartis built its strategy on trust with healthcare providers, developing the ‘Day One’ access scheme to guarantee reimbursement of the initial list price and future rebates in line with country-specific HTA assessments. The scheme was a clear signal from Novartis regarding their negotiation flexibility to meet market-specific needs. In addition, by making concessions, Novartis could expand the middle ground of negotiations by convincing healthcare systems of the curative value of Zolgensma so that agreements could be reached.
“Cell and gene therapies (CGTs) have heralded a new era of medicine, promising curative treatments for patients with chronic and often fatal diseases”
However, flexible in pricing negotiations is only one half of the equation; pharma must also demonstrate a commitment to continue generating RWE as a supporting evidence base following the approval of their drug. Marking a watershed moment for reimbursement, Germany’s G-BA demanded R WE for Zolgensma, stating that “medicines are being approved earlier and earlier with increasingly weaker evidence”. (3) The limited evidence available for initial assessment by Germany’s IQWiG (7) meant that Novartis is obligated to conduct a registry trial to support ongoing HTAs. (4) This should serve as a cautionary tale to pharma that their strategy for long-term RWE generation must be built in alliance with HTAs, not only to facilitate reimbursement but to defend the franchise against competition.
A Framework Redefining Stakeholder Dynamics
Finding common ground between pharma and healthcare systems is key to unlocking access for patients. Alignment on the evidence requirements for CGTs is essential. Both pharma and healthcare systems must engage early, clarify equitable expectations, and assess the feasibility and timing of evidence-generation efforts to find common ground to work together. With the evidence at hand, determining the value of CGTs is the next step in demonstrating clinical and societal benefits. Agreement on the curative potential of a CGT product must be established to ensure pricing models are adapted to reflect value for all stakeholders. Our proposed framework (Figure 2) requires pharma and healthcare systems to embed changes in ways of working that can reduce the investment risk for all parties and, most importantly, expedite patient access.
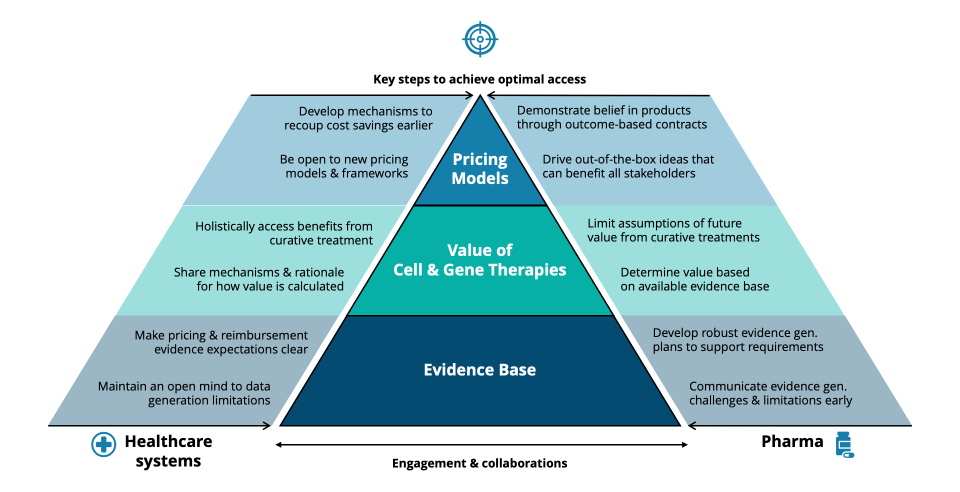
Figure 2: Framework to support pharma and healthcare systems achieve improved access for new CGTs.
Establishing a solid evidence base lays the foundations for all price negotiations. Unlike traditional drug products, where pivotal data is often sufficient evidence for market approval and reimbursement, CGTs require the generation of robust evidence before, during, and after market approval to demonstrate value to patients and payers. For pharma, openly sharing clinical trial designs and future evidence generation plans from an early stage can allow a more robust model for developing a solid evidence base. In tandem, healthcare systems can provide evidence-based expectations on P&R requirements early on. However, the payer expectations of having a complete dataset with hard endpoints at launch will likely need to shift, given the long-term curative potential of CGTs. Both parties must have an open mind on the limitations and challenges of data generation for these unique therapies. In practice, we envisage the functionality of third-party evidence management, involving stakeholders from pharma, government, and academia, to facilitate the standardized cross-border collection, management, and distribution of data. Initiatives such as the European Health Data Evidence Network (EHDEN) and H ematology Outcomes Network in Europe (HONEUR), as well as existing cross-border HTA alliances such as Beneluxa initiative, EUnetHTA, and FINOSE, could be used as a vehicle to implement a standardized RWE framework across multiple geographies. Ultimately, communicating and settling on a mutual understanding of where the evidence produced by pharma and evidence threshold from healthcare systems match up will set the groundwork for smooth value and pricing negotiations.
Following agreement on the evidence base, alignment of the intrinsic value and curative potential of CGT products is required. If deep discounts are a prerequisite to further negotiation, the dynamic will never be mutually beneficial. In Zolgensma’s case, Novartis deemed the therapy to be worth $2 million for its one-time curative effect. With less than five years of market surveillance data, Zolgensma’s life-long curative potential has many more years to be realized; what if the clinical benefits and socio-economic cost-savings are less than initially thought? Pharma will be restrained from negotiating the value of CGTs based on their unique selling point of long-term savings through curative action. Focusing on evidence generation will be critical; however, pharma and healthcare systems must define a common framework to effectively assess each CGT’s value and potential savings, which is currently missing. One option for pharma could be to determine CGT value based on the available evidence at launch with additional outcome-based contracts capturing the incremental value of curative potential through further evidence generation. In response, healthcare systems can be more open in their value calculations while incorporating a holistic view of CGT benefits within the health system. The benefits of curative treatments go beyond treating the target patient; broader societal and economic value can be reaped from improved productivity of treated patients and their caregivers, relief of consultation and hospitalization costs borne by the health system, and more. Whether the potential of curative treatments should be assessed for a life-long period or for a shorter time horizon (e.g., 10 years), this is something for pharma and healthcare systems to agree upon.
“Pharma and healthcare systems must unite and take drastic collaborative action to ready global healthcare systems for the new wave of emerging CGTs or face hindered patient access”
Open communication of evidence and value culminates in the ability to craft tailored pricing models advantageous for pharma and healthcare systems, acting to move all stakeholders into the sacred middle ground. Applying innovative pricing models to CGTs is critical for the access ecosystem; however, these can come in various forms and must be tailored to product value recognition and accepted by healthcare systems. In the context of price negotiations, the increased trust fostered through collaborative and transparent engagements in creating an evidence and value base enables aligned pricing strategies to be developed. To expand access opportunities, pharma and healthcare systems need to be open to new ideas, understand each other’s needs, and deepen collaboration at each step of driving towards a common goal of bringing new CGTs to patients. Healthcare systems need to be reassured that they will receive the expected value for the treatments they pay for, while pharma needs guarantees that their investments will be duly rewarded.
Before robust aligned evidence, unified value recognition, and tailored pricing models can drive CGT access in the future, the current short-term price-focused mindset must be reformed, or it will continue to act as a barrier to innovation. Removing this roadblock will take time, but concerted efforts by pharma can catalyze this. In addition, pharma should take a more active role in shaping policy towards a system that encourages long-term investments and cost savings for healthcare systems from curative therapies. In short, the dynamic between healthcare systems and pharma must be redefined, as they are both playing for the same team where the end goal can save countless patients’ lives. Sharing risks, building trust, and working together – can pharma and healthcare systems embrace change and meet in the middle to succeed in achieving the true potential of CGTs?
Embracing change, accessing value – we all stand to win from CGTs
Innovation has unlocked value for society time and time again. For CGTs to positively impact patients’ lives with the true potential envisioned, drastic shifts are needed from pharma and healthcare systems. Changing evidence expectations, increasing collaboration between key stakeholders, and flexibility in adopting new pricing frameworks are vital steps to achieving access and unlocking the maximal value of CGTs.
Historically, pharma and healthcare systems have been able to navigate complex challenges to bring new innovations to market collectively; CGTs are set to be the next life-changing chapter in the healthcare journey. The future holds opportunities for significant rewards for all stakeholders within the space as more CGT products progress ever closer to market approval, with the full effect of treating broader indications yet to be seen. Pharmaceutical companies that are quick to acknowledge the challenges and embrace the critical success factors identified will win in this exciting space. Though the required changes take time and involve multiple steps, pharma should not hold back from investing early in these efforts to prepare healthcare systems for widespread CGT access and to ensure the successful launch of their promising pipelines. With optimized access, the true benefits of CGTs can be reaped by pharma, healthcare systems, and most importantly, patients – all stand to win.


